Charge transport in conductive polymers and nanochannels for neuromorphic applications
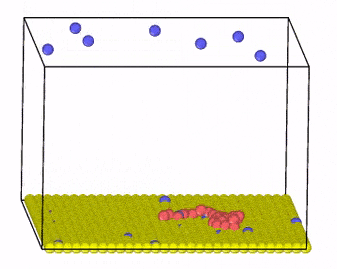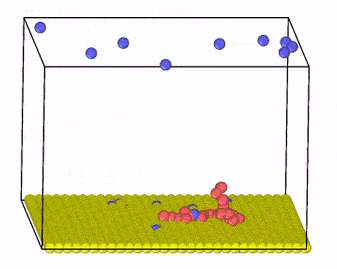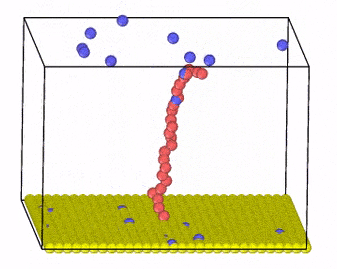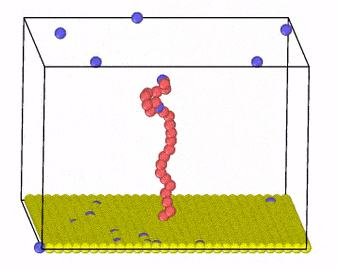
We investigate these systems by employing Monte Carlo and molecular dynamics simulations, combined with new algorithm development for charge transfer within classical MD simulations. Moreover, we develop methods that incorporate hydrodynamic interactions with coarse-grained models of charged molecules and soft materials.
Designing Self Assembly of Lipid Nanoparticles
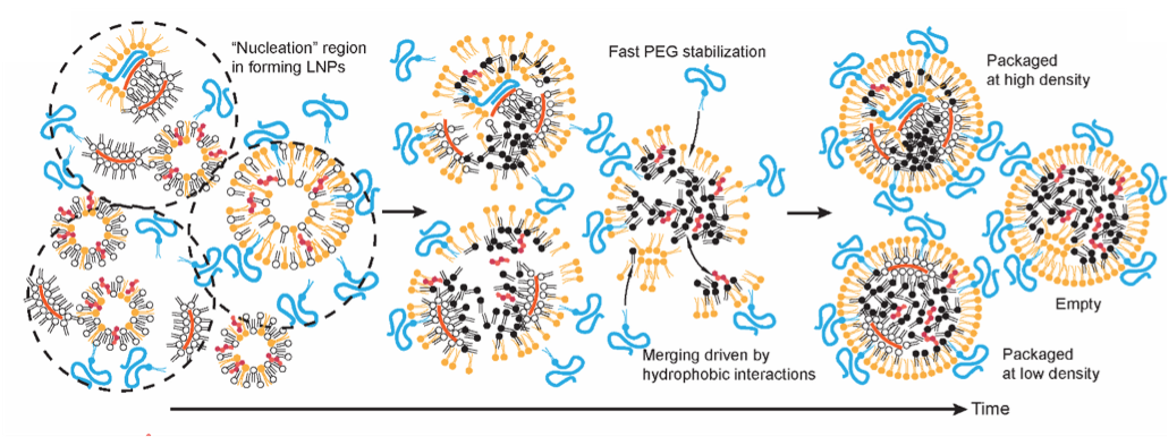
Lipid nanoparticle self-assembly and growth. We strive to understand the kinetics of RNA-lipid nanoparticle self-assembly, how to kinetically control the particle size and minimize polydispersity. We mainly employ analytical kinetic theory, kinetic Monte Carlo and molecular dynamics simulations. Ongoing collaboration with the group of Hai-Quan Mao.
S. Li, Y. Hu*, J. Lin, Z. Schneiderman, F. Shao, L. Wei, A. Li, K. Hsieh, E. Kokkoli, T. Curk*, H.-Q. Mao*, T.-H. Wang*
ACS Nano 2024, 18, 24, 15729–15743 [pdf]
Designing Organic electrochemical transistor biosensors
Many medical conditions can be detected or diagnosed by analyzing chemical compounds circulating in the bloodstream or that are in other body liquids.To confidently detect these proteins quickly and easily, electronic sensors are needed that signal when they are in contact with the proteins that indicate the medical condition of interest to a patient. One way to improve the certainty that a sensor signal is indicating the presence of a particular protein is to provide circuits that remove signals that come from other compounds in the liquids being analyzed, like salt, fats, and additional proteins. This project investigates a new circuit, constructed with new biologically-derived electronic materials, designed to remove these unwanted signals that make it more difficult to detect the proteins that indicate medical conditions. In collaboration with Howard Katz and Supported by NSF CBET award No. 2402407.
Y. Song, N. Chen, T. Curk*, H. E. Katz*, Molecules 29 (7), 1459 (2024)
Charge regulation of polyelectrolytes, nanoparticles and nanochannels
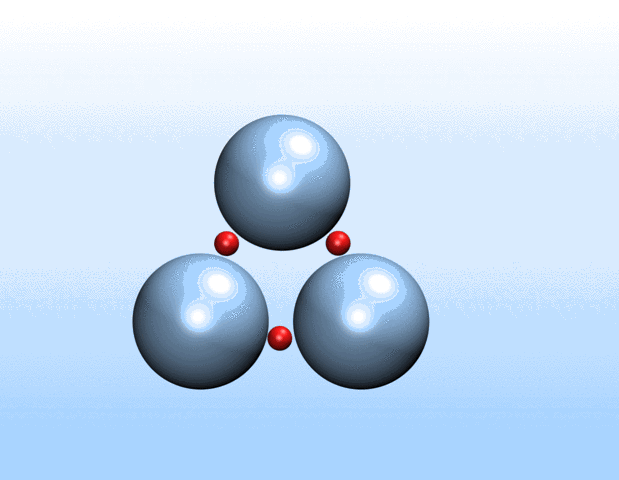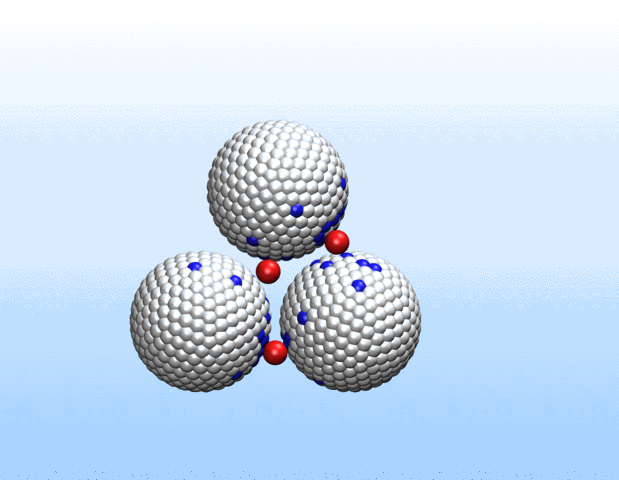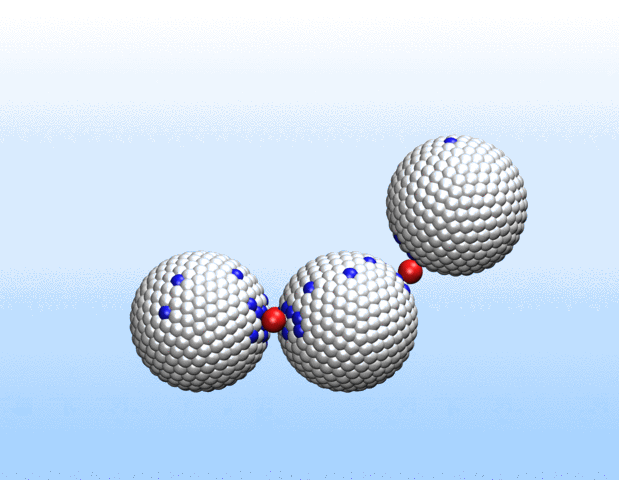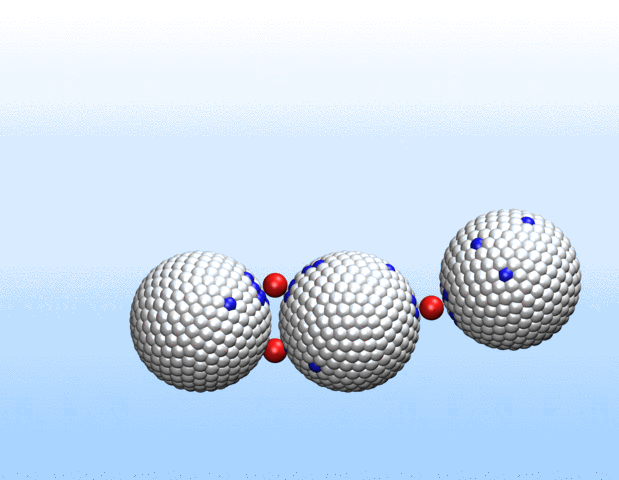
We are investigaing charge-structure coupling in nanoparticles and polyelectrolytes for designing e.g. nano-actuators for soft robotics. Nanoparticles, polyelectrolytes and biomolecules in solution acquire charge through the dissociation or association of surface groups. Thus, a proper description of their electrostatic interactions requires sampling of protonation states and the use of charge-regulating boundary conditions rather than the commonly employed constant-charge approximation. We provide an open-source implementation of the charge-regulation solver for the LAMMPS molecular dynamics package.
Optimal packing of polymers (DNA) in viral capsids.
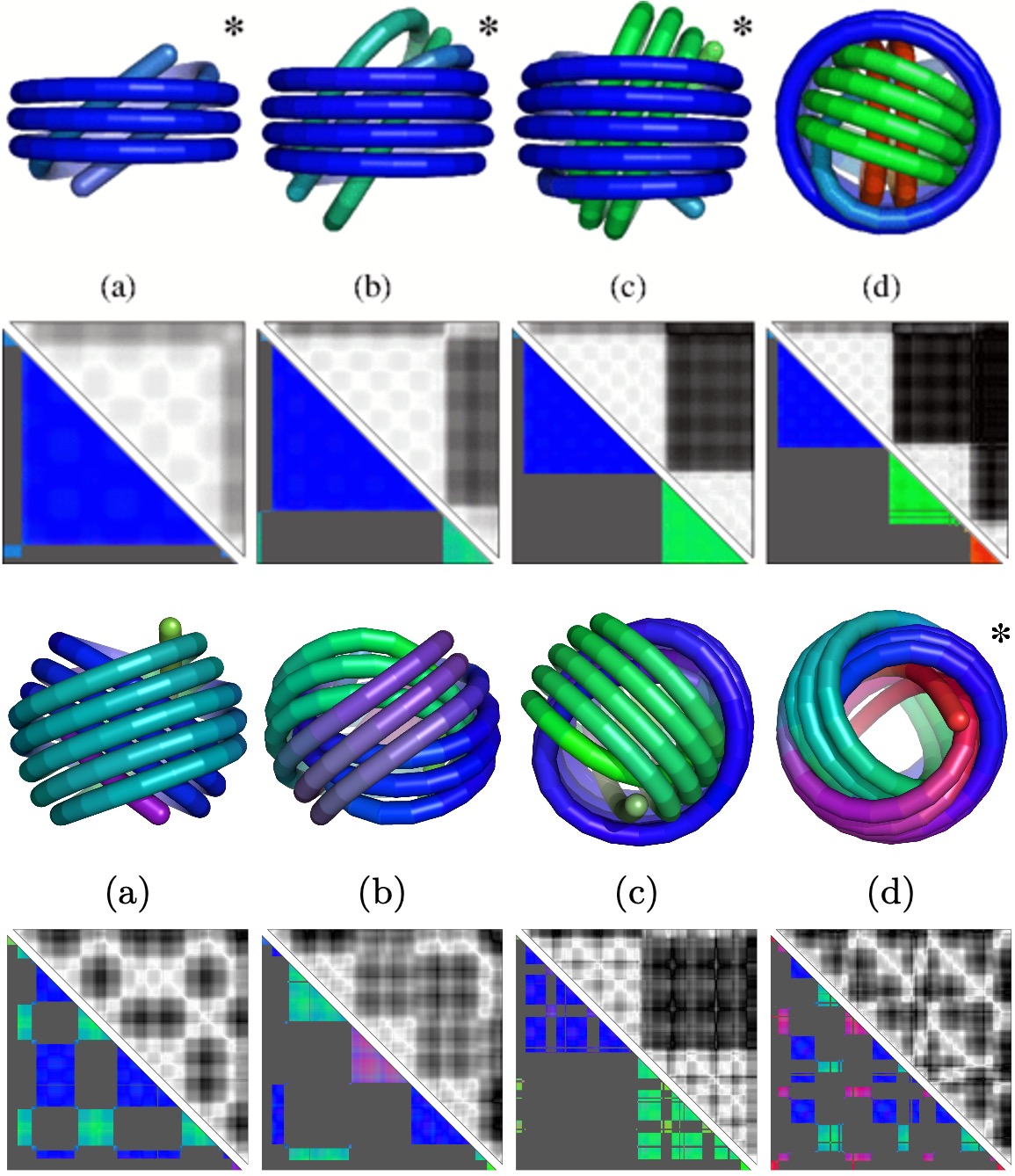
The packing of DNA and dsRNA is crucial in viral assembly and DNA delivery. Using analytical calculations and MD simulations we show that DNA spontanously forms an ordering mosaic of multiple homogeneously ordered domains. We observe concentric spools, topological links and spool-nematic ordering.
Designing superselective targeting in multivalent polymers
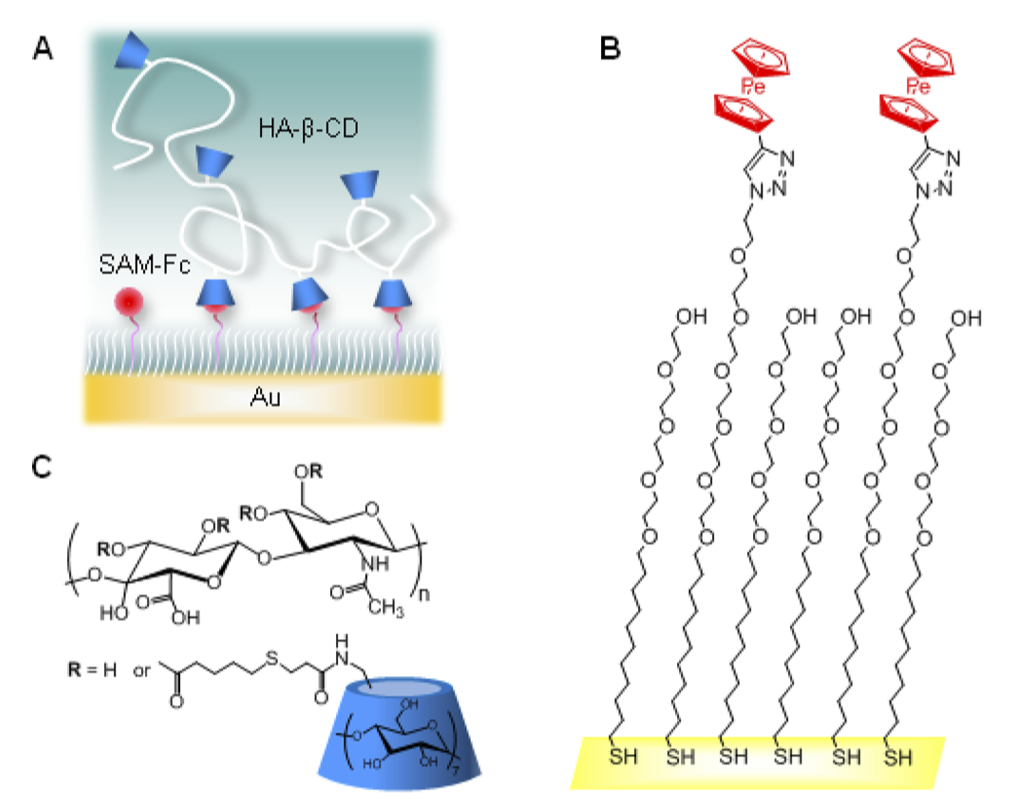
We rationalized design rules for super-selective targeting using multivalent polymers and particles, in collaboration with experimental groups of Dr. Galina Dubacheva at ENS Paris-Saclay and Prof. Ralf Richter at Leeds. We have also improved the sequence design of nucleotide probes for pathogen genome detection. Currently, our focus is on understanding the kinetics of multivalent interactions to design selective drug delivery vehicles for cell membrane targeting.
G. V. Dubacheva, T. Curk, R. Auzly-Velty, D. Frenkel, and R. P. Richter
Proc. Natl. Acad. Sci. U.S.A. 112, 5579–5584 (2015)
Designing multivalent interactions

We investigate interactions between nanoparticles and multicomponent membranes and membrane trafficking. We also show the effects of intrinsic curvature of receptors, and uncover how multivalent DNA--peptide aggregates can trigger immune response via activation of TLR9, leading to autoimmune disorders. Current focus is on investigating how tuning the receptor--receptor interactions and receptor clustering in the membrane can be used to control multivalent binding and endocytosis.
T. Curk, J. Dobnikar, and D. Frenkel
Proc. Natl. Acad. Sci. U.S.A. 114, 7210–7215 (2017)
T. Curk P. Wirnsberger, J. Dobnikar, D. Frenkel, and A. Šarić
Nano Lett. 18, 9 (2018) [front cover]
N. W. Schmidt, F. Jin, R. Lande, T. Curk, W. Xian, C. Lee, L. Frasca, D. Frenkel, J. Dobnikar, M. Gilliet, and G. C. L. Wong
Nature Materials 14, 696–700 (2015)